- lauraclark849
- Mar 24
- 34 min read
Updated: Apr 14
Technical Review Article | Open Access | Published 26th March 2026
Functional Silver and Gold Nanoparticles in Pharmaceutical Technology: A Deep Review of Formulation Techniques
Mahesh Kumar Posa¹ᵃ, Ravi Kumar Kota², V.T.Iswariya³, P.Moulika⁴, S.Gejalakshmi⁵, Jagdish Kumar Arun¹ᵇ, Seema Sharma⁶ , and Sampat Singh Tanwar⁶*, Ravi Kumar Kota², V.T.Iswariya³, P.Moulika⁴, S.Gejalakshmi⁵, Jagdish Kumar Arun¹ᵇ, Seema Sharma⁶ , and Sampat Singh Tanwar⁶* | EJPPS | 311 (2026) https://doi.org/10.37521/ejpps31102
Back to Journals | Article |Abstract| References | Authors
Abstract
Nanotechnology has transformed contemporary pharmaceuticals by providing nanoscale materials that enable precise drug delivery, controlled release, and targeted therapeutic applications. Silver nanoparticles (AgNPs) and gold nanoparticles (AuNPs) have emerged as exceptionally versatile agents owing to their adjustable size, shape, surface characteristics, and biocompatibility. This review offers an extensive examination of their pharmaceutical potential, emphasizing synthesis techniques namely, chemical, biological green, and physical methods and their impact on nanoparticle morphology, stability, and functionalization. Silver nanoparticles (AgNPs) exhibit significant antimicrobial efficacy and are extensively utilized in wound healing and infection management, whereas gold nanoparticles (AuNPs) are favoured in oncology, gene delivery, photothermal therapy, and diagnostics owing to their inertness and optical characteristics. Surface engineering techniques, including PEGylation and ligand conjugation (e.g., antibodies, aptamers, folic acid), enhance systemic circulation, targeting precision, and therapeutic efficacy. The review examines drug loading methodologies utilizing covalent and non-covalent interactions, as well as hybrid delivery systems such as liposomes and hydrogels, which facilitate improved entrapment efficiency and targeted delivery. Mechanistic understanding of drug release through diffusion, swelling, erosion, and degradation is underpinned by mathematical models such as Higuchi, Korsmeyer–Peppas, and zero-order kinetics. Recent innovations, including stimuli-responsive systems, microneedle patches, and AI-assisted formulation optimization are also examined. Notwithstanding substantial advancements, critical obstacles persist in large-scale production, nanotoxicity assessment, and regulatory authorization. Nonetheless, AgNPs and AuNPs signify a transformative advancement in pharmaceutical development, possessing the capacity to propel the forthcoming generation of personalized, adaptive, and intelligent drug delivery systems.
Keywords: Silver nanoparticles (AgNPs) , gold nanoparticles (AuNPs); biological green, gene delivery.
Introduction
The advancement of nanotechnology has initiated a profound shift in pharmaceutical sciences by enabling the creation of nanoscale drug delivery systems that provide enhanced therapeutic precision prolonged drug release and reduced systemic toxicity through highly controlled physicochemical design and biological responsiveness. Among the extensive range of nanoscale materials investigated for biomedical use silver nanoparticles and gold nanoparticles have gained significant attention because they possess distinctive physicochemical features excellent biological tolerance and remarkable functional versatility that allow the development of next generation therapeutic systems.¹⁻³ These metallic nanostructures support site specific drug targeting, varied modes of drug loading and tightly regulated drug release through engineered surface functionalities and environmentally responsive mechanisms that adapt to physiological stimuli. Silver based nanoparticles exhibit powerful antimicrobial and antifungal activity against resistant pathogens while gold-based nanoparticles serve in cancer therapy photothermal ablation imaging diagnostics and gene delivery due to their optical and electronic characteristics and their inert chemical nature. Advances in materials science now allow the engineering of nanoparticle size, shape, surface charge and colloidal stability within the sub-one hundred nanometer range which significantly improves pharmacokinetic performance biodistribution and biological interactions.⁴,⁵ Their synthesis can be achieved through controlled chemical reduction high energy physical methods or green biological routes that utilize plant extracts or microbial agents and that align with principles of ecological safety and biocompatibility. In pharmaceutical formulations silver and gold nanoparticles are frequently modified with polymers such as PEG PLGA or chitosan to improve colloidal stability circulation time and immune system evasion whereas active targeting ligands including antibodies aptamers and folate analogues are employed to achieve selective tissue accumulation. Drug release from metallic nanoparticles proceeds through diffusion erosion swelling or degradation-based processes that respond to changes in pH enzymatic activity or thermal variation and these mechanisms are commonly analyzed using mathematical kinetic models such as zero order first order Higuchi and Korsmeyer Peppas to predict and optimize therapeutic behavior under physiological conditions. Through the integration of stimulus responsive architectures rational surface modification and predictive kinetic modeling silver and gold nanoparticles have become highly adaptable platforms that support targeted therapy, sustained drug release, imaging guided intervention and innovative nanomedicine strategies.⁶,⁷
The present review therefore seeks to provide a complete and critical scientific examination of silver and gold nanoparticles in pharmaceutical research with emphasis on their synthesis techniques, formulation strategies, drug loading mechanisms and drug release kinetics with the goal of establishing a cohesive understanding of the key physicochemical principles that control their therapeutic performance. This review surveys chemical physical and biologically mediated synthesis routes to outline how structural and functional attributes are regulated during nanoparticle fabrication and it explores formulation principles required to generate stable biocompatible and high performance metallic nanocarriers. It further presents an in-depth evaluation of the kinetic models used to describe the release of therapeutic agents from nanoscale systems in order to clarify the mechanisms that determine clinical efficacy. In addition this review discusses the broad pharmaceutical applications of silver and gold nanoparticles in antimicrobial therapy oncology immunotherapy and targeted delivery alongside an analysis of safety issues, regulatory expectations and ethical considerations that govern their translational potential. Finally this work identifies the major scientific challenges, unresolved research gaps and emerging opportunities that will guide future investigations in transforming silver and gold nanoparticle platforms into clinically reliable scalable and therapeutically powerful systems for modern pharmaceutical innovation.
2. A Beginning to Nanoparticles of Silver and Gold (AgNPs & AuNPs)
2.1 Physicochemical Properties
Silver nanoparticles (AgNPs) exhibit high surface reactivity, antimicrobial potential, and conductivity, but face challenges related to stability and toxicity. In contrast, gold nanoparticles (AuNPs) offer superior chemical inertness, biocompatibility, and tuneable optical properties, making them excellent platforms for drug delivery, diagnostics, and photothermal therapy.⁸ The selection between AgNPs and AuNPs depends on the desired application, with AgNPs being preferred for antimicrobial use and AuNPs for targeted therapy and imaging.
Table 1 - Physicochemical Properties of Silver Nanoparticles (AgNPs) and Gold Nanoparticles (AuNPs)
Property | Silver Nanoparticles (AgNPs) | Gold Nanoparticles (AuNPs) |
Chemical Composition | Elemental silver (Ag⁰) or silver oxide (Ag₂O) | Elemental gold (Au⁰) |
Size Range | Typically 1–100 nm | Typically 1–100 nm |
Shape Variability | Spheres, rods, cubes, prisms, and wires | Spheres, rods, stars, cages, and shells |
Surface Charge (Zeta Potential) | Usually negative; can be tuned using capping agents | Can be positive, negative, or neutral based on functionalization |
Surface Reactivity | High; prone to oxidation and Ag⁺ ion release | Low; chemically inert and highly stable |
Surface Functionalization | Coated with stabilizers (e.g., PVP, citrate, chitosan) | Functionalized via thiol (-SH) linkages, PEG, antibodies |
Optical Property | Exhibits localized surface plasmon resonance (LSPR) | Exhibits strong surface plasmon resonance (SPR) |
Plasmon Resonance Peak | ~400–450 nm (depends on size/shape) | ~520–550 nm (depends on size/shape) |
Electrical Conductivity | High; useful in conductive inks and biosensors | High; used in electrochemical sensors |
Solubility and Stability | Less stable; prone to aggregation and photo-oxidation | Highly stable; excellent colloidal and photostability |
Toxicity | Moderate to high (dose-dependent cytotoxicity and ROS generation) | Low toxicity and highly biocompatible |
Antimicrobial Activity | Strong intrinsic antimicrobial action | Weak antimicrobial unless functionalized |
Biodegradability | Limited; ion release can affect biological systems | Poorly biodegradable but inert and safe under most conditions |
Color in Solution | Yellow-brown to grey | Ruby red to purple (depending on size and aggregation) |
2.2 Mechanism of Action in Biological Systems
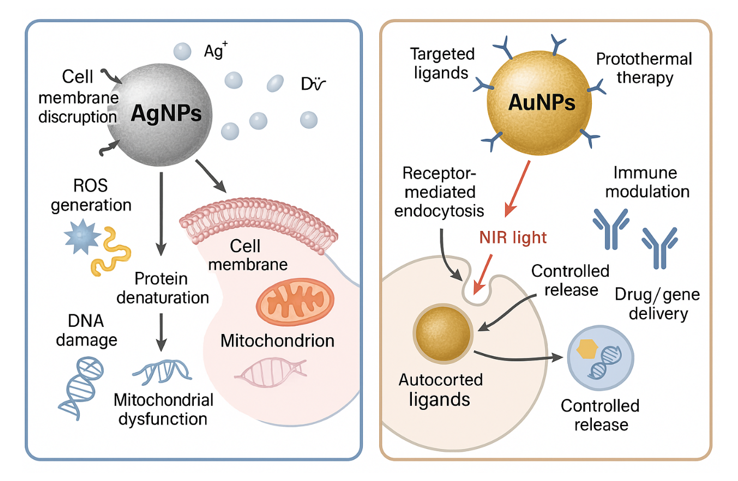
Silver nanoparticles (AgNPs) exert their biological effects primarily through the release of silver ions (Ag⁺) which disrupt cell membranes, generate reactive oxygen species (ROS), interfere with enzyme activity by binding to thiol groups, and cause DNA damage, collectively leading to cell death particularly in microbial systems. They also induce mitochondrial dysfunction and oxidative stress in mammalian cells contributing to cytotoxicity. In contrast gold nanoparticles (AuNPs) are biologically inert and act mainly as carriers for drugs, genes, or imaging agents.⁹ Their mechanism involves receptor-mediated endocytosis, targeted payload delivery, and stimulus-responsive release under specific pH or redox conditions. AuNPs also exhibit photothermal properties via surface plasmon resonance (SPR), enabling them to convert light into localized heat for cancer cell ablation, while their ability to enhance immune responses makes them suitable as vaccine adjuvants and diagnostic tools.
3. Formulation Techniques of AgNPs and AuNPs
3.1 Chemical Reduction Methods
Chemical reduction is among the most widely adopted and foundational techniques for synthesizing silver (AgNPs) and gold nanoparticles (AuNPs) in pharmaceutical sciences due to its cost-effectiveness, reproducibility and capacity for size and shape control. In this method, metal precursors such as silver nitrate (AgNO₃) or chloroauric acid (HAuCl₄) are reduced to their metallic states using chemical reducing agents in the presence of stabilizing or capping agents which serve to prevent aggregation and control nanoparticle growth.¹⁰ Sodium borohydride (NaBH₄), ascorbic acid and trisodium citrate are the most commonly employed reducing agents. NaBH₄ being a strong reductant facilitates rapid nucleation and results in the formation of small, monodisperse AgNPs with diameters often less than 10 nm. However due to the high reactivity of this agent, immediate stabilization using capping agents such as polyvinylpyrrolidone (PVP), polyethylene glycol (PEG) or citrate is essential to avoid uncontrolled growth and particle agglomeration.¹¹ In contrast ascorbic acid serves as a mild and biocompatible reducing agent often employed in synthesizing both AgNPs and AuNPs when slower nucleation and larger particle sizes are desired. It enables better morphological control and is frequently used in biomedical formulations where lower cytotoxicity is prioritized.¹² Trisodium citrate is notable for its dual role as both a reducing and stabilizing agent and forms the basis of the well-established Turkevich method for synthesizing spherical gold nanoparticles. In this classical approach HAuCl₄ is reduced under boiling conditions to form AuNPs typically in the size range of 10–30 nm, and the resulting citrate-capped nanoparticles exhibit remarkable colloidal stability and ease of surface functionalization.¹³ Numerous studies have demonstrated how reaction conditions including temperature, pH, molar ratios of reagents, and stirring speed significantly influence the properties of the resulting nanoparticles. For example, increasing the temperature can enhance the reduction kinetics and promote uniform particle size whereas variations in pH can alter the ionization of stabilizing agents thus affecting the colloidal stability and morphology of the nanoparticles.¹⁴ Higher pH values generally favour smaller nanoparticles due to the increased nucleation rate and enhanced activity of citrate ions. Several researchers have utilized these parameters to tailor nanoparticles for specific pharmaceutical applications. For instance, Song and Kim (2009) synthesized highly stable AgNPs using NaBH₄ and PVP achieving uniform spherical particles with average diameters of approximately 5 nm, suitable for antimicrobial formulations.
Huang et al. (2007) employed ascorbic acid and cetyltrimethylammonium bromide (CTAB) to synthesize triangular and hexagonal gold nanoplates, demonstrating the possibility of morphology control through mild reduction pathways. Beyond basic synthesis post-synthetic surface functionalization is integral to the application of these nanoparticles in drug delivery.¹⁵ Gold nanoparticles in particular, possess a strong affinity for thiol (-SH) groups, enabling efficient conjugation with targeting ligands, drugs, peptides, and nucleic acids. This feature supports their widespread use in theragnostic where simultaneous drug delivery and diagnostic imaging are required.¹⁶ Surface modification with PEG (PEGylation) is another commonly used strategy to improve nanoparticle biocompatibility, circulation time, and resistance to opsonization making chemically synthesized AuNPs especially suitable for intravenous administration in oncology.¹⁷ Morphological control also extends to non-spherical shapes such as rods, stars, and cages, which can be synthesized via seed-mediated growth techniques in the presence of CTAB and reducing agents like ascorbic acid. These non-spherical nanoparticles offer altered surface plasmon resonance (SPR) properties, making them ideal candidates for photothermal therapy especially in cancer treatment.¹⁸ Chemical reduction methods also lend themselves to hybrid formulations where nanoparticles are encapsulated within or conjugated to liposomes, micelles or polymeric carriers enhancing the payload capacity and enabling controlled drug release.
Table 2: Representative Studies on Chemical Reduction Based Synthesis of AgNPs and AuNPs
Study | Nanoparticle | Reducing Agent | Stabilizer | Size/Shape | Application |
Song & Kim (2009) | AgNP | NaBH₄ | PVP | ~5 nm (spherical) | Antimicrobial |
Huang et al. (2007) | AuNP | Ascorbic Acid | CTAB | Plates/Triangles | Photothermal therapy |
Grabar et al. (1995) | AuNP | Citrate | Citrate | 10–30 nm spheres | Immunoassays |
Sharma et al. (2009) | AuNP | Citrate | Citrate | 15–20 nm | SPR tuning |
Jana et al. (2001) | AuNP | Ascorbic Acid | CTAB | Rods (AR 2–5) | Optical imaging |
Ghosh et al. (2008) | AuNP | NaBH₄ | PEG | ~20 nm | Drug delivery (cancer) |
3.2 Green Synthesis Techniques
Green synthesis of metallic nanoparticles has emerged as a sustainable, eco-friendly and biocompatible alternative to conventional chemical and physical methods for producing silver (AgNPs) and gold nanoparticles (AuNPs). This approach leverages biological resources such as plant extracts, microbial cultures, enzymes and natural polymers to reduce metal ions to nanoparticles while simultaneously acting as capping agents that stabilize the synthesized particles.¹⁹ Unlike traditional methods that often employ toxic chemicals such as sodium borohydride or cetyltrimethylammonium bromide (CTAB) green synthesis ensures minimal environmental impact and improved biocompatibility of the resulting nanoparticles.²⁰ In plant-mediated synthesis phytochemicals such as flavonoids, alkaloids, tannins, terpenoids and phenolic acids play pivotal roles in reducing metal salts (e.g., AgNO₃ for AgNPs and HAuCl₄ for AuNPs) and stabilizing the nanoparticles. These biomolecules provide functional groups that not only initiate nucleation but also prevent agglomeration resulting in nanoparticles with controlled morphology and enhanced biological activity. ²¹ Numerous studies have demonstrated the successful green synthesis of AgNPs and AuNPs using diverse plant species such as Azadirachta indica (neem), Camellia sinensis (green tea), Moringa oleifera, Ocimum sanctum, and Terminalia arjuna each contributing unique phytoconstituents that influence particle size, shape, and stability. For example, Ahmed et al. (2016) reported the synthesis of AgNPs using Moringa oleifera leaf extract polyphenols and amino acids served as reducing and capping agents, producing spherical nanoparticles (~10–30 nm) with potent antimicrobial properties. In a similar vein, Philip (2010) utilized cinnamon extract for the synthesis of AuNPs and demonstrated its efficacy in producing stable monodisperse particles suitable for photothermal applications.
The process typically involves mixing a plant extract with an aqueous metal salt solution under ambient conditions and observing a colour change due to surface plasmon resonance (SPR) which signifies nanoparticle formation. The morphology of the nanoparticles can be modulated by adjusting factors such as extract concentration, pH, temperature, and incubation time. Beyond plants, microbial-mediated synthesis using bacteria (Bacillus subtilis, Pseudomonas aeruginosa), fungi (Fusarium oxysporum, Aspergillus niger) and algae (Chlorella vulgaris, Sargassum wightii) offers another promising avenue for green nanoparticle synthesis. ²² Microbial cells or their supernatants contain enzymes, peptides and reducing cofactors such as NADH and nitrate reductase which facilitate the bioreduction of metal ions to nanosized particles. For example, Kalishwaralal et al. (2010) synthesized AuNPs using Bacillus licheniformis culture filtrate, producing biocompatible nanoparticles with antiangiogenic activity suitable for cancer therapy. ²³ Fungal-mediated synthesis, in particular, is advantageous due to the extracellular secretion of large quantities of reducing enzymes and proteins making downstream processing and nanoparticle recovery easier. Microbial synthesis suffers from limitations such as slow growth rates, potential pathogenicity, and the need for sterile conditions, which restrict its scalability and industrial viability. ²⁴ To overcome these challenges, hybrid green methods combining plant extracts with microbial metabolites or incorporating enzymatic systems are under exploration to achieve synergistic benefits in nanoparticle synthesis. ²⁵ The morphology of green-synthesized nanoparticles is highly tuneable and ranges from spherical to triangular, hexagonal, and rod-like structures, depending on the source of the biological reductant and the synthesis conditions. ²⁶ Morphological control is critical because the shape and size of nanoparticles influence their surface plasmon resonance (SPR), cellular uptake, drug loading capacity, and biological activity. For instance, triangular AuNPs exhibit sharper SPR peaks and enhanced photothermal conversion efficiency compared to spherical ones making them suitable for imaging and therapeutic applications. ²⁷ In the pharmaceutical domain, green-synthesized AgNPs and AuNPs have shown significant promise as antimicrobial agents, drug carriers, anticancer agents and diagnostic tools. ²⁸ Their inherent biological activity, particularly the broad-spectrum antimicrobial effect of AgNPs, is enhanced by the bioactive compounds embedded on their surface during green synthesis. ²⁹ These surface-bound phytochemicals not only stabilize the particles but also synergize with metal ions to disrupt microbial cell walls, inhibit DNA replication, and induce oxidative stress. On the other hand, green-synthesized AuNPs have been effectively conjugated with anticancer drugs, peptides and targeting ligands, enabling selective tumour targeting for enhanced bioavailability, and reduced systemic toxicity. For example, Shukla et al. (2012) demonstrated the use of neem-derived AuNPs for delivering doxorubicin to breast cancer cells, achieving improved cytotoxicity with minimal impact on healthy cells. The absence of synthetic surfactants or chemical modifiers makes green nanoparticles particularly attractive for oral, topical, and injectable pharmaceutical formulations. Characterization of green-synthesized nanoparticles requires advanced analytical tools such as UV-Vis spectroscopy, dynamic light scattering (DLS), transmission electron microscopy (TEM), Fourier transform infrared spectroscopy (FTIR), and X-ray diffraction (XRD) to confirm particle size, morphology, crystallinity, and functional groups.
Table 3: Summary of Green Synthesis Methods for Silver and Gold Nanoparticles
Biological Source | Type | Reducing/Capping Agent | Nanoparticle Size & Shape | Application |
Moringa oleifera (leaf extract) | Plant | Polyphenols, flavonoids | AgNPs, 10–30 nm, spherical | Antimicrobial |
Cinnamomum zeylanicum (bark) | Plant | Polyphenols, terpenoids | AuNPs, 20–40 nm, spherical | Photothermal therapy |
Azadirachta indica (neem) | Plant | Alkaloids, tannins | AuNPs, 15–50 nm, spherical/triangular | Drug delivery (anticancer) |
Fusarium oxysporum (fungus) | Fungi | Enzymes, NADH, reductase | AgNPs, ~25 nm, spherical | Wound healing, antimicrobial |
Bacillus licheniformis | Bacteria | Extracellular proteins, enzymes | AuNPs, 10–20 nm, spherical | Anti-angiogenic (cancer therapy) |
Chlorella vulgaris (algae) | Algae | Pigments, amino acids | AgNPs, 20–50 nm, spherical/hexagonal | Antibacterial and biosensing |
3.3 Biological Synthesis
Biological synthesis of silver (AgNPs) and gold nanoparticles (AuNPs) involves a series of eco-friendly and biocompatible processes utilizing living organisms such as bacteria, fungi, algae, and viruses. ³⁰ The process begins with the preparation of metal salt solutions like AgNO₃ or HAuCl₄, which are then mixed with biological agents or their extracts. ³¹ These biological entities produce enzymes, proteins, and metabolites such as NADH-dependent nitrate reductase in bacteria or electron shuttle quinones in fungi that facilitate the reduction of metal ions (Ag⁺, Au³⁺) into metallic nanoparticles. ³² The reduction occurs either intracellularly within the microbial or fungal cells, or extracellularly in the culture medium. Biomolecules secreted by these organisms act as both reducing and capping agents, initiating nanoparticle nucleation and stabilizing their size, shape, and dispersion. Parameters such as pH, temperature, extract concentration, and incubation time are fine-tuned to control nanoparticle morphology and yield.³³ Following synthesis, nanoparticles are recovered via centrifugation or filtration, especially in extracellular approaches. ³⁴ The synthesized nanoparticles are characterized using advanced techniques like UV-Vis spectroscopy, FTIR, TEM, DLS, and XRD to confirm size, shape, and surface functionality. ³⁵ These biogenic nanoparticles are then applied in antimicrobial coatings, drug delivery, cancer therapy, and diagnostic biosensors. To improve reproducibility and scalability, processes such as microbial cell immobilization, hybrid phyto-microbe systems, and genetic engineering of high-yield microbial strains are employed. ³⁶ Lastly, biosynthesized nanoparticles undergo rigorous toxicity testing, biodistribution studies, and regulatory compliance to meet Good Manufacturing Practices (GMP) for clinical applications.
Table 4: Summary of biological synthesis methods for Silver and Gold Nanoparticles
Biological Organism | Type | Mechanism | NP Properties | Application | Reference |
Bacillus licheniformis | Bacteria | Extracellular enzyme (nitrate reductase) | AgNPs, 20–50 nm, spherical | Antimicrobial | Natarajan et al., 2010 |
Pseudomonas aeruginosa | Bacteria | Protein-mediated reduction | AuNPs, 10–25 nm, stable | Biosensing | Jain et al., 2011 |
Fusarium oxysporum | Fungi | Quinones, extracellular enzymes | AgNPs, ~25 nm, uniform | Wound healing, antimicrobial | Durán et al., 2005 – |
Aspergillus fumigatus | Fungi | Intracellular accumulation | AuNPs, 15–40 nm, spherical | Drug delivery | Bhainsa & D'Souza, 2006 – |
Chlorella vulgaris | Algae | Pigment-mediated extracellular synthesis | AuNPs, 20–60 nm, stable | Gene delivery | Dahoumane et al., 2016 – |
Tobacco mosaic virus | Virus | Bio-template for anisotropic growth | AuNPs, rods/discs, aligned | Nanoassembly, imaging | Lee et al., 2002 – |
3.4 Physical Methods
Physical methods for synthesizing AgNPs and AuNPs are top-down approaches that fragment bulk metals into nanoparticles using mechanical or thermal energy, without involving chemical reducing or stabilizing agents. ³⁷ Key techniques include laser ablation, evaporation condensation, UV irradiation, and spark discharge.³⁸ In laser ablation, high-energy laser pulses target a metal immersed in a liquid medium, dislodging atoms that nucleate into nanoparticles; parameters such as wavelength, pulse duration, and laser–target distance control particle size. ³⁹ Evaporation–condensation involves vaporizing bulk metal in a furnace and condensing the vapour into nanoparticles in an inert gas or vacuum environment, yielding highly crystalline, pure particles. ⁴⁰ UV irradiation employs ultraviolet light to photochemically reduce metal ions in aqueous or polymer media to form nanoparticles, offering a green alternative though with slower kinetics. These methods produce ligand-free, high-purity nanoparticles with minimal contamination, making them suitable for sensitive applications, although challenges such as high equipment costs, scalability limitations, and lack of shape control persist. ⁴¹ Hybrid physical methods, such as laser–chemical combinations and plasma synthesis, are being explored to enhance yield, uniformity, and surface stabilization.
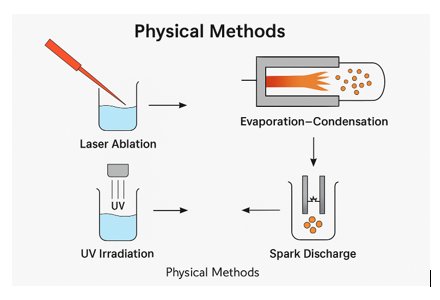
Table No 5: Physical Synthesis Methods for Silver and Gold Nanoparticles
Technique | Nanoparticle Type | Synthesis Conditions | Nanoparticle Properties | Advantages | Limitations | Reference |
Laser Ablation | AuNPs, AgNPs | Pulsed laser on metal target in water/ethane | 10–30 nm, spherical, high purity | Chemical-free, stable colloids, tunable sizes | Expensive setup, low throughput | Amendola & Meneghetti, 2009 |
Evaporation–Condensation | AuNPs | Thermal vaporization in inert gas; condensation on cool zone | Crystalline, narrow size distribution | Pure particles, high crystallinity | Energy-intensive, vacuum required | Kim et al., 2007 |
UV Irradiation | AgNPs, AuNPs | UV exposure of metal salt solution in polymer matrix | 15–25 nm, stable in PVA matrix | Low cost, green method, ambient temperature | Slow process, limited control over size | Mallick et al., 2001 |
Microwave Irradiation | AgNPs, AuNPs | Dielectric heating of metal salt solution | Rapid synthesis, spherical/irregular shapes | Fast reaction time, energy efficient | Hot spots, uneven size distribution | Polte et al., 2010 |
Spark Discharge | AgNPs | High-voltage discharge between metal electrodes | Small (~10 nm), crystalline nanoparticles | No surfactants, highly pure particles | Low yield, expensive apparatus | Tsuji et al., 2005 |
3.5 Polymeric and Lipid-Based Approaches
The preparation of polymeric and lipid-based nanoparticle formulations involves either the direct encapsulation of AgNPs or AuNPs within biodegradable polymer matrices such as PLGA, PEG, chitosan, or PVA, or surface modification of the nanoparticles using biopolymers through techniques like PEGylation. ⁴² These polymers form protective shells around the nanoparticles to prevent aggregation and enable co-delivery of both hydrophilic and hydrophobic drugs.⁴³ In PEGylation, PEG chains are covalently attached to the nanoparticle surface to prolong systemic circulation and enhance tumour targeting via the EPR effect. Similarly, chitosan coating is used for mucoadhesive and antimicrobial applications.⁴⁴ Lipid-based methods involve incorporating AgNPs or AuNPs into liposomes, SLNs, or nanostructured lipid carriers (NLCs), where nanoparticles are either entrapped in the aqueous core or embedded within the lipid bilayer. In liposome-based preparations, functional agents such as folic acid may be added to target cancer cells, and thermosensitive liposomes are designed for drug release upon infrared stimulation. ⁴⁵ Polymer-lipid hybrid nanoparticles (PLHNs) are also formulated by combining polymeric cores with lipid shells, offering improved stability, drug loading, and sustained release.⁴⁶ These systems may be engineered to respond to external stimuli such as pH, enzymes, or heat, enabling site-specific drug delivery.

4. Drug Loading and Encapsulation Approaches
Nanoparticles composed of silver (AgNPs) and gold (AuNPs) have gained considerable attention as drug delivery vehicles due to their unique physicochemical properties, including tuneable surface chemistry, high surface area-to-volume ratio, and ease of functionalization. ⁴⁷ A critical aspect in the development of such nanocarriers is the approach used to load and encapsulate therapeutic agents. Efficient drug loading not only determines the therapeutic payload but also affects the release kinetics, bioavailability, and systemic circulation time.⁴⁸ This section provides an in-depth comparative analysis of drug loading strategies onto AgNPs and AuNPs, with specific emphasis on covalent versus non-covalent interactions, entrapment efficiency, and formulation stability.
4.1 Covalent vs Non-Covalent Interactions
The method of drug attachment to nanoparticles significantly influences drug release profiles, bio-distribution, and targeting capability. ⁴⁹ Broadly drug-nanoparticle interactions can be classified into covalent and non-covalent linkages.
Covalent Interactions
Covalent conjugation involves the formation of stable chemical bonds between the drug molecule and functional groups on the nanoparticle surface.⁵⁰ For instance, thiol (-SH) groups present on biomolecules can form strong covalent bonds with the gold surface due to Au–S affinity. Similarly, carboxyl or amine-functionalized AgNPs can be conjugated with drugs via EDC/NHS coupling chemistry. ⁵¹ This approach ensures high drug stability and controlled release due to bond cleavage under specific physiological conditions (e.g., pH, enzymatic activity, or redox conditions).
Non-Covalent Interactions
Non-covalent strategies rely on weaker forces such as electrostatic interactions, hydrogen bonding, van der Waals forces, and hydrophobic interactions.⁵² This method is advantageous for maintaining the structural integrity and bioactivity of labile drugs.⁵³ For example, positively charged doxorubicin can electrostatically interact with citrate-stabilized negatively charged AuNPs. Similarly, hydrophobic drugs such as paclitaxel can be loaded onto PEGylated AgNPs through hydrophobic interactions.
Table 6 Comparative Summary Covalent vs Non-Covalent Interactions
Feature | Covalent Interaction | Non-Covalent Interaction |
Stability | High | Moderate to Low |
Drug Release Control | Excellent | Rapid or burst release |
Impact on Drug Bioactivity | Possible loss of activity | Retains structural integrity |
Synthesis Complexity | High (requires functionalization) | Low (simple adsorption) |
Reversibility | Irreversible | Reversible |
4.2 Entrapment Efficiency
Entrapment efficiency (EE%) refers to the percentage of drug successfully incorporated into or onto nanoparticles compared to the total amount initially used during formulation, and it plays a vital role in determining dosing frequency, therapeutic efficacy, and cost-effectiveness of nanoparticle-based drug delivery systems.⁵⁴ Various factors influence EE%, including the solubility and charge of the drug. Hydrophilic drugs often show lower EE% due to repulsion from similarly hydrophilic nanoparticle surfaces, while hydrophobic or amphiphilic drugs exhibit better entrapment. ⁵⁵ The surface chemistry of nanoparticles, particularly the presence of functional groups such as carboxyl, amine, or thiol, affects the drug's binding affinity, while the size and surface area of nanoparticles also play a role, with smaller particles offering greater interaction surfaces for drug binding. ⁵⁶ Additionally, formulation conditions such as pH and ionic strength can influence electrostatic interactions, especially in non-covalent drug loading.⁵⁷ Silver nanoparticles (AgNPs) typically show higher EE%, especially when combined with polymeric coatings such as chitosan, PEG, or PVA, which provide a matrix for drug entrapment. For instance, PVA-coated AgNPs have demonstrated EE% values of 75–85% for tetracycline.⁵⁸ Conversely, gold nanoparticles (AuNPs), due to their chemically inert surfaces, tend to have lower EE% in non-covalent formulations, but they achieve excellent EE% values (80–95%) when drugs are covalently attached using linker chemistries such as PEG-thiol or mercaptoundecanoic acid, making them highly effective for targeted and sustained drug delivery applications.
Table No 7 Comparative Insight Entrapment Efficiency
Parameter | Silver NPs | Gold NPs |
Typical EE% (non-covalent) | 60–85% | 40–70% |
EE% with covalent loading | 70–90% | 80–95% |
EE Modulation Strategies | Polymer coating | Surface functionalization |
Best Applications | Antibacterial, wound healing | Cancer therapy, gene delivery |
4.3 Stability Considerations
Stability of the drug–nanoparticle complex is crucial to maintain shelf-life, therapeutic efficacy, and batch-to-batch reproducibility, as instability can result in nanoparticle aggregation, premature drug release, or degradation of biological activity. ⁵⁹ Physical stability concerns primarily revolve around aggregation, which is common in metallic nanoparticles due to van der Waals forces; this is often mitigated using surface coatings such as polyethylene glycol (PEG), polyvinylpyrrolidone (PVP), or surfactants such as Tween 80 to provide steric hindrance. ⁶⁰ Gold nanoparticles (AuNPs) generally offer superior colloidal stability owing to their strong surface plasmon resonance (SPR), which reduces the likelihood of aggregation. ⁶¹ In contrast, silver nanoparticles (AgNPs), being more chemically reactive, are vulnerable to oxidation and the uncontrolled release of silver ions, which can destabilize the system; encapsulation in biocompatible polymers or the use of mild reducing agents such as ascorbic acid helps enhance their stability. ⁶² Chemical stability is another critical factor, especially in covalently linked drug–nanoparticle systems where bond cleavage can occur under varying pH conditions or due to enzymatic activity, compromising the integrity of the conjugate. ⁶³ Non-covalent systems are even more fragile, with a higher risk of drug desorption in complex biological environments rich in proteins, potentially leading to reduced therapeutic performance and increased systemic toxicity.
Table No 8 Comparative Stability
Stability Parameter | Silver NPs | Gold NPs |
Colloidal Stability | Moderate (requires coating) | High (especially PEGylated) |
Chemical Stability | Prone to oxidation | Chemically inert |
Storage Stability | Up to 3–6 months (coated) | 6–12 months (covalent) |
pH Sensitivity | High | Moderate |
5. Drug Release Kinetics
Drug release kinetics is a crucial determinant of therapeutic efficacy, bioavailability, and systemic safety in nanoparticulate drug delivery systems. ⁶⁴ For silver (AgNPs) and gold nanoparticles (AuNPs), the mechanisms of drug release and their kinetic modeling are key factors influencing design, formulation, and clinical performance.⁶⁵ This section explores the fundamental mechanisms governing drug release from AgNPs and AuNPs, highlights various kinetic models used for evaluation, and discusses controlled and targeted release strategies for optimizing therapeutic outcomes.
5.1. Mechanisms of Drug Release
Drug release from nanoparticles is governed by key mechanisms diffusion, erosion, swelling, and degradation, each influenced by the drug-nanoparticle interaction, encapsulation strategy, and physiological environment. Diffusion predominates in systems where drugs are adsorbed via weak interactions, as seen in PEGylated gold nanoparticles (AuNPs) and chitosan-coated silver nanoparticles (AgNPs), allowing gradual release through polymer pores or surface ligands. ⁶⁶ Erosion-based release occurs in biodegradable or hybrid constructs, such as polymer-AuNP composites or chitosan-AgNP matrices, particularly under acidic or enzymatically active conditions. ⁶⁷ Swelling-driven release is characteristic of hydrogel-embedded or stimuli-responsive nanoparticles, where aqueous-induced expansion generates diffusion channels, enabling site-specific delivery in response to pH or temperature shifts. ⁶⁸ Degradation, although limited in inert metals such ass Au and Ag, becomes relevant when surface-bound organic components degrade or under oxidative stress, with AgNPs releasing silver ions that contribute to both antimicrobial action and drug liberation. ⁶⁹ These mechanisms often act concurrently, and their dominance depends on the physicochemical properties of the system and the target microenvironment.

5.2. Mathematical Models for Kinetics
To analyse drug release profiles from silver and gold nanoparticle-based systems, several mathematical models are employed to elucidate the kinetics and underlying mechanisms of drug liberation. ⁷⁰ Zero-order kinetics, characterized by a constant release rate independent of drug concentration, is observed in some sustained-release platforms such as lipid-coated AgNPs and hydrogel-embedded AuNPs, although it remains challenging in metallic systems due to surface-mediated release dynamics. ⁷¹ First-order kinetics, where the release rate is concentration-dependent, typically describes systems with surface-adsorbed drugs such as PVP-stabilized AgNPs or AuNPs with labile linkers. ⁷² The Higuchi model, based on Fickian diffusion, effectively interprets release from polymer-embedded matrices by correlating the released amount with the square root of time.⁷³ The Korsmeyer–Peppas model, a semi-empirical equation, captures more complex release behaviours involving multiple mechanisms especially in stimuli-responsive formulations—by analysing the release exponent (n) to differentiate between Fickian, anomalous, and case-II transport.⁷⁴ Each model provides insights tailored to specific nanoparticle configurations: zero-order for consistent release, first-order for rapid desorption, Higuchi for matrix-controlled systems, and Korsmeyer–Peppas for hybrid or responsive systems, thereby aiding in the design and prediction of therapeutic performance under physiological conditions.
6. Pharmaceutical Applications
Silver (AgNPs) and gold nanoparticles (AuNPs) offer a broad range of pharmaceutical applications due to their distinctive physicochemical properties and biocompatibility. ⁷⁷ AgNPs are widely recognized for their strong intrinsic antibacterial and indirect antiviral properties through reactive oxygen species (ROS) generation, making them ideal for use in wound dressings, antibacterial coatings, and antimicrobial gels. ⁷⁸ Though their use in cancer therapy is limited by potential toxicity, AgNPs have demonstrated pro-apoptotic effects in cancer cells and are being explored in combination therapies. ⁷⁹ In contrast, AuNPs are extensively used in cancer therapeutics due to their excellent biocompatibility, tuneable surface chemistry, and utility in photothermal therapy, drug delivery, and imaging. ⁸⁰ Both AgNPs and AuNPs find applications in dermatology—AgNPs in FDA-approved wound healing products and AuNPs in anti-aging and inflammatory skin formulations.⁸¹ For oral drug delivery, AgNPs require enteric or biopolymer coatings to reduce gastrointestinal toxicity, while AuNPs exhibit higher stability and enhanced absorption for bioactives such as curcumin. Parenterally, AuNPs excel in targeted cancer treatment due to prolonged circulation, whereas AgNPs are studied for sepsis therapies.⁸² In transdermal systems, AgNPs are included in antimicrobial patches, while AuNPs are incorporated into microneedles and nanogels for painless vaccine and drug delivery.⁸³ AuNPs also lead in vaccine and immunotherapy applications, functioning as adjuvants and antigen carriers, whereas AgNPs are limited due to cytotoxicity but are under investigation for mucosal vaccine use.⁸⁴ Furthermore, AuNPs are widely used in diagnostic imaging techniques such as CT and MRI and serve as efficient gene delivery carriers for plasmid DNA and siRNA.⁸⁵ Both nanoparticle types have also demonstrated anti-inflammatory effects, with AuNPs showing strong cytokine modulation useful in chronic inflammatory diseases.⁸⁶ This diverse application spectrum positions AgNPs and AuNPs as powerful agents in next-generation therapeutics and personalized medicine.
Table No 9 Pharmaceutical Applications
Application Area | Silver Nanoparticles (AgNPs) | Gold Nanoparticles (AuNPs) |
Antibacterial and Antiviral Agents | High antibacterial; indirect antiviral (via ROS); cytotoxicity at high doses; used in wound dressings, coatings | Moderate antibacterial (with functionalization); high antiviral potential with siRNA or drugs |
Cancer Therapeutics | Exhibit anti-cancer effects via apoptosis and ROS; less common due to toxicity | Effective in targeted therapy, photothermal therapy; conjugated with ligands for EPR effect |
Wound Healing and Dermatological Uses | Used in FDA-approved wound dressings (e.g., Acticoatâ„¢); moderate anti-inflammatory effect | Used in anti-aging and anti-inflammatory skin formulations; aids in collagen synthesis |
Oral Formulations | Limited use; requires protective coatings (e.g., alginate, chitosan) to reduce GI toxicity | Stable, biocompatible; used with drugs like curcumin and quercetin for enhanced absorption |
Parenteral Formulations | Used in antimicrobial IV formulations; risk of cytotoxicity at higher doses | Effective in cancer diagnostics and treatment; long circulation times |
Transdermal Systems | Commonly used in antimicrobial gels and patches | Used in microneedle-based and nanogel formulations for drug/vaccine delivery |
Vaccines and Immunotherapy | Limited use; potential as pro-inflammatory adjuvants in mucosal vaccines | Well-studied as adjuvants; tested with peptide/DNA antigens; safe and effective T-cell activation |
Imaging and Diagnostics | Occasional use in imaging due to SPR effect; less stable than AuNPs | Widely used in diagnostic imaging (CT, MRI, SPR) due to optical properties and surface functionality |
Gene Delivery | Exploratory studies on gene loading via electrostatic interaction; concerns over stability | Efficient carriers for plasmid DNA, siRNA, and miRNA; low cytotoxicity and high transfection efficiency |
Anti-inflammatory Therapy | Demonstrated anti-inflammatory effects in wound and skin care products | Promising results in reducing cytokines (e.g., TNF-α, IL-6); used in rheumatoid arthritis models |
7. Challenges and Future Prospects
Despite the remarkable advances in silver (AgNPs) and gold nanoparticles (AuNPs) for pharmaceutical applications, several critical challenges continue to impede their widespread clinical adoption.⁸⁷ One major hurdle is the difficulty in scaling up nanoparticle production while maintaining consistent physicochemical properties such as size, shape, surface charge, and functionalization.⁸⁸ Batch-to-batch variability, high production costs especially for AuNPs and the need for advanced stabilization strategies for AgNPs hinder reliable large-scale manufacturing.⁸⁹ Meeting regulatory standards under Good Manufacturing Practices (GMP) further complicates scale-up, requiring strict quality assurance protocols.⁹⁰ The future of nanoparticle production may lie in microfluidic reactors and continuous flow systems that ensure sterility and reproducibility, complemented by real-time monitoring using Process Analytical Technologies (PAT). ⁹¹ Personalized nanomedicine also offers promising avenues for tailored therapies, with AuNPs functionalized by targeting ligands enabling precise drug delivery based on patient-specific biomarkers.⁹² Integration with pharmacogenomics can help select appropriate nanoparticle formulations based on genetic profiles, but this requires advanced diagnostics, clinical support tools, and bioinformatics infrastructure—making implementation complex and expensive.⁹³ Meanwhile, concerns surrounding nanotoxicology and long-term safety are pressing.⁹⁴ AgNPs, despite their strong antimicrobial properties, can induce oxidative stress, cytotoxicity, and DNA damage, with risks of tissue accumulation and even argyria upon prolonged exposure.⁹⁵ AuNPs are more biocompatible but still face scrutiny over organ retention, immune response, and incomplete biodegradation.⁹⁶ Environmental toxicity from nanoparticle waste adds another layer of concern. To mitigate such risks, the development of biodegradable and self-degrading nanoparticles, in vivo imaging tools, and organ-on-chip toxicity models is essential.⁹⁷ Artificial intelligence (AI) is emerging as a powerful solution for many of these issues, enabling predictive modeling, in silico screening, and digital twin simulations to optimize nanoparticle design and personalize therapy.⁹⁸ AI and machine learning models can fine-tune formulation parameters for optimal biological performance while reducing reliance on traditional trial-and-error methods.⁹⁹ Despite current limitations such as data standardization and regulatory hesitancy, the integration of AI, robotics, and automation heralds a new era in nanopharmaceutical development.¹⁰⁰ Ultimately, the convergence of nanotechnology and AI offers transformative potential to overcome current barriers ushering in a future of scalable, safe, and personalized nanoparticle-based therapies across oncology, infectious diseases, immunotherapy, and regenerative medicine.
Conclusion
Silver nanoparticles (AgNPs) and gold nanoparticles (AuNPs) have become versatile and highly effective agents in pharmaceutical nanotechnology, due to their adjustable physicochemical properties, biocompatibility, and ability for multifunctional drug delivery. This review emphasizes that both nanoparticles can be synthesized through chemical, green, biological, and physical methods, each facilitating precise control over size, morphology, and surface properties. Silver nanoparticles (AgNPs) demonstrate significant antimicrobial effectiveness and are extensively utilized in wound healing and infection management, while gold nanoparticles (AuNPs) are superior in targeted cancer treatment, gene delivery, imaging, and photothermal applications owing to their inert properties and surface plasmon resonance. Functionalization methods, such as PEGylation and ligand conjugation with antibodies, aptamers, or folic acid, improve nanoparticle circulation duration, stability, and targeting precision. Covalent and non-covalent drug loading strategies, frequently integrated with hybrid carriers like liposomes and hydrogels, enhance entrapment efficiency, therapeutic efficacy, and targeted release. The release kinetics, influenced by mechanisms such as diffusion, erosion, swelling, and degradation, are accurately represented by established mathematical models including Higuchi, zero-order, and Korsmeyer–Peppas, thereby enhancing formulation predictability and clinical design.
Despite the clear biomedical potential of AgNPs and AuNPs, obstacles such as aggregation, oxidative degradation (in the case of AgNPs), elevated production costs, and restricted biodegradability (pertaining to AuNPs) continue to impede large-scale clinical application. Furthermore, the necessity for standardized manufacturing protocols, nanotoxicity assessments, and regulatory coherence remains. Recent innovations to surmount these obstacles encompass stimuli-responsive systems (pH, redox, enzyme-triggered release), microneedle patches, and intelligent hydrogel composites. The incorporation of artificial intelligence (AI) and in silico predictive modeling into nanoparticle design aims to enhance formulation variables, predict biological responses, and facilitate precision therapy via digital twin simulations. In conclusion, silver and gold nanoparticles signify a transformative advancement in nanomedicine by facilitating controlled, responsive, and targeted drug delivery systems. Their ongoing advancement, supported by interdisciplinary methodologies and sophisticated computational instruments, is set to transform contemporary therapeutics and establish new benchmarks for personalized pharmaceutical care.
References
1. Aguilar-Garay, R., Martínez-Castro, S., Sánchez-Vázquez, M., & Contreras-Torres, F. F. (2024). Silver and gold nanoparticles as multifunctional platforms for antimicrobial and therapeutic applications. Pharmaceuticals, 17(9), 1134. https://doi.org/10.3390/ph17091134
2. Sati, A., Kumar, P., Rawat, R., & Tewari, D. (2025). Silver nanoparticles for cancer therapy and drug-delivery applications: Comprehensive insights into synthesis and biomedical functions. ACS Omega, 10(3), 5124–5141. https://doi.org/10.1021/acsomega.4c11045
3. Amalkar, S. V., Hatwar, P. R., Bakal, R. L., & Kohale, N. B. (2024). Advances in gold nanoparticle-mediated drug-delivery systems. GSC Biological and Pharmaceutical Sciences, 28(3), 169–179. https://doi.org/10.30574/gscbps.2024.28.3.0324
4. Hosny, S., El-Shinawy, M., & El-Damak, D. (2025). Silver nanoparticles in drug-delivery systems: Biomedical properties challenges and future prospects. Applied Nanoscience. https://doi.org/10.1007/s13204-025-03091-9
5. Tunç, T., Acar, T., & Ocakoglu, K. (2024). Synthesis and characterization of drug-loaded silver nanoparticles for antimicrobial and anticancer drug delivery. Cancer Nanotechnology, 14, Article 12. https://doi.org/10.1186/s12645-023-00243-1
6. Laib, I., Soudani, N., & Hadj-Ziane-Zafour, A. (2025). Tailoring innovative silver nanoparticles for modern pharmaceutical use: Stability surface functionalization and therapeutic potential. Nanomedicine: Nanotechnology Biology and Medicine. https://doi.org/10.1016/j.nano.2025.103927
7. Peng, L., Zhang, Y., Chen, H., & Li, Z. (2025). Emerging trends in nanoparticle-based drug delivery systems with focus on inorganic and hybrid nanocarriers. Nanoscale, 17(4), 1521–1540. https://doi.org/10.1039/D4NR05114A
8. 8 Islam, S., Rahman, M., & Choudhury, S. (2025). Nanoparticle-mediated targeted drug delivery: Design considerations current challenges and future directions. Nano Today, 60, 102129. https://doi.org/10.1016/j.nantod.2025.102129
9. Sabzehali, S., Nakhjavani, M., & Hosseini, S. (2025). Metal nanoparticles as multifunctional carriers in drug-delivery systems with emphasis on silver and gold nanostructures. Egyptian Journal of Chemistry, 68(2), 301–320. https://doi.org/10.21608/ejchem.2024.25609.6025
10. Meher, A., Rout, P. K., & Jena, S. (2024). Applications of silver nanoparticles in medicine: An updated review on therapeutic potentials and safety considerations. Materials Today Chemistry, 36, 102521. https://doi.org/10.1016/j.mtchem.2024.102521
11. Baptista, P.V., McCusker, M.P., Carvalho, A., Ferreira, D.A., Mohan, N.M., Martins, M. and Fernandes, A.R., 2018. Nano-strategies to fight multidrug resistant bacteria—“A Battle of the Titans”. Frontiers in Microbiology, 9, p.1441. https://doi.org/10.3389/fmicb.2018.01441
12. Chudasama, B., Vala, A.K., Andhariya, N., Upadhyay, R.V. and Mehta, R.V., 2010. Enhanced antibacterial activity of bifunctional Fe₃O₄–Ag core–shell nanostructures. Nano Research, 3(8), pp.516–527. https://doi.org/10.1007/s12274-010-1037-z
13. Daniel, M.C. and Astruc, D., 2004. Gold nanoparticles: assembly, supramolecular chemistry, quantum-size-related properties, and applications toward biology, catalysis, and nanotechnology. Chemical Reviews, 104(1), pp.293–346. https://doi.org/10.1021/cr030698+
14. Dykman, L. and Khlebtsov, N., 2012. Gold nanoparticles in biomedical applications: recent advances and perspectives. Chemical Society Reviews, 41(6), pp.2256–2282. https://doi.org/10.1039/c1cs15166e
15. Fratoddi, I., Venditti, I., Cametti, C. and Russo, M.V., 2015. How toxic are gold nanoparticles? The state-of-the-art. Nano Research, 8(6), pp.1771–1799. https://doi.org/10.1007/s12274-014-0697-3
16. González-Ballesteros, N., Rodríguez-González, J.B. and Rodríguez-Argüelles, M.C., 2017. Green synthesis of gold nanoparticles using Chamaemelum nobile extract: effect on the proliferation of human tumor cells. Journal of Drug Delivery Science and Technology, 42, pp.263–271. https://doi.org/10.1016/j.jddst.2017.04.015
17. Iravani, S., Korbekandi, H., Mirmohammadi, S.V. and Zolfaghari, B., 2014. Synthesis of silver nanoparticles: chemical, physical and biological methods. Research in Pharmaceutical Sciences, 9(6), pp.385–406.
18. Korsmeyer, R.W., Gurny, R., Doelker, E., Buri, P. and Peppas, N.A., 1983. Mechanisms of solute release from porous hydrophilic polymers. International Journal of Pharmaceutics, 15(1), pp.25–35. https://doi.org/10.1016/0378-5173(83)90064-9
19. Rai, M., Yadav, A. and Gade, A., 2009. Silver nanoparticles as a new generation of antimicrobials. Biotechnology Advances, 27(1), pp.76–83. https://doi.org/10.1016/j.biotechadv.2008.09.002
20. Sharma, V.K., Yngard, R.A. and Lin, Y., 2009. Silver nanoparticles: Green synthesis and their antimicrobial activities. Advances in Colloid and Interface Science, 145(1-2), pp.83–96. https://doi.org/10.1016/j.cis.2008.09.002
21. Sperling, R.A., Rivera Gil, P., Zhang, F., Zanella, M. and Parak, W.J., 2008. Biological applications of gold nanoparticles. Chemical Society Reviews, 37(9), pp.1896–1908. https://doi.org/10.1039/B712170A
22. Gurunathan, S., Park, J.H., Han, J.W. and Kim, J.H., 2015. Comparative assessment of the apoptotic potential of silver nanoparticles synthesized by Bacillus tequilensis and Calocybe indica in MDA-MB-231 human breast cancer cells: targeting p53 for anticancer therapy. International Journal of Nanomedicine, 10, pp.4203–4222. https://doi.org/10.2147/IJN.S81450
23. Iravani, S., 2011. Green synthesis of metal nanoparticles using plants. Green Chemistry, 13(10), pp.2638–2650. https://doi.org/10.1039/C1GC15386B
24. Jain, P.K., Huang, X., El-Sayed, I.H. and El-Sayed, M.A., 2006. Noble metals on the nanoscale: optical and photothermal properties and some applications in imaging, sensing, biology, and medicine. Accounts of Chemical Research, 39(9), pp.738–745. https://doi.org/10.1021/ar050280
25. Kim, J.S., Kuk, E., Yu, K.N., Kim, J.H., Park, S.J., Lee, H.J., Kim, S.H., Park, Y.K., Park, Y.H., Hwang, C.Y. and Kim, Y.K., 2007. Antimicrobial effects of silver nanoparticles. Nanomedicine: Nanotechnology, Biology and Medicine, 3(1), pp.95–101. https://doi.org/10.1016/j.nano.2006.12.001
26. Zhou, Y., Kong, Y., Kundu, S., Cirillo, J.D. and Liang, H., 2012. Antibacterial activities of gold and silver nanoparticles against Escherichia coli and Bacillus Calmette-Guérin. Journal of Nanobiotechnology, 10(1), p.19. https://doi.org/10.1186/1477-3155-10-19
27. Khlebtsov, N. and Dykman, L., 2010. Biodistribution and toxicity of engineered gold nanoparticles: a review of in vitro and in vivo studies. Chemical Society Reviews, 40(3), pp.1647–1671. https://doi.org/10.1039/C0CS00018C
28. Huang, X., Jain, P.K., El-Sayed, I.H. and El-Sayed, M.A., 2008. Plasmonic photothermal therapy (PPTT) using gold nanoparticles. Lasers in Medical Science, 23(3), pp.217–228. https://doi.org/10.1007/s10103-007-0470-x
29. Murphy, C.J., Gole, A.M., Hunyadi, S.E. and Orendorff, C.J., 2006. One-dimensional colloidal gold and silver nanostructures. Inorganic Chemistry, 45(19), pp.7544–7554. https://doi.org/10.1021/ic060190x
30. Zhang, X.D., Wu, D., Shen, X., Liu, P.X., Fan, F.Y. and Fan, S.J., 2012. In vivo renal clearance, biodistribution, toxicity of gold nanoclusters. Biomaterials, 33(18), pp.4628–4638. https://doi.org/10.1016/j.biomaterials.2012.03.022
31. Thanh, N.T.K. and Green, L.A., 2010. Functionalisation of nanoparticles for biomedical applications. Nano Today, 5(3), pp.213–230. https://doi.org/10.1016/j.nantod.2010.05.003
32. Mody, V.V., Siwale, R., Singh, A. and Mody, H.R., 2010. Introduction to metallic nanoparticles. Journal of Pharmacy and Bioallied Sciences, 2(4), pp.282–289. https://doi.org/10.4103/0975-7406.72127
33. Ahmed, S., Saifullah, Ahmad, M., Swami, B.L. and Ikram, S., 2016. Green synthesis of silver nanoparticles using Azadirachta indica aqueous leaf extract. Journal of Advanced Research, 7(1), pp.17–28. https://doi.org/10.1016/j.jare.2015.02.007
34. McShan, D., Ray, P.C. and Yu, H., 2014. Molecular toxicity mechanism of nanosilver. Journal of Food and Drug Analysis, 22(1), pp.116–127. https://doi.org/10.1016/j.jfda.2014.01.010
35. Xie, J., Lee, S. and Chen, X., 2010. Nanoparticle-based theranostic agents. Advanced Drug Delivery Reviews, 62(11), pp.1064–1079. https://doi.org/10.1016/j.addr.2010.07.009
36. Ramalingam, B., Parandhaman, T. and Das, S.K., 2016. Antimicrobial properties and mechanism of action of silver nanoparticles synthesized from Litchi chinensis fruit peel extract. Materials Science and Engineering: C, 68, pp.456–465. https://doi.org/10.1016/j.msec.2016.06.020
37. Jain, D., Daima, H.K., Kachhwaha, S. and Kothari, S.L., 2009. Synthesis of plant-mediated silver nanoparticles using Papaya fruit extract and evaluation of their antimicrobial activities. Digest Journal of Nanomaterials and Biostructures, 4(3), pp.557–563.
38. Song, J.Y. and Kim, B.S., 2009. Rapid biological synthesis of silver nanoparticles using Plantago asiatica extract. Bioprocess and Biosystems Engineering, 32(1), pp.79–84. https://doi.org/10.1007/s00449-008-0224-6
39. Huang, X., Neretina, S. and El-Sayed, M.A., 2009. Gold nanorods: from synthesis and properties to biological and biomedical applications. Advanced Materials, 21(48), pp.4880–4910. https://doi.org/10.1002/adma.200802789
40. Grabar, K.C., Freeman, R.G., Hommer, M.B. and Natan, M.J., 1995. Preparation and characterization of Au colloid monolayers. Analytical Chemistry, 67(4), pp.735–743. https://doi.org/10.1021/ac00101a002
41. Jana, N.R., Gearheart, L. and Murphy, C.J., 2001. Wet chemical synthesis of silver nanorods and nanowires of controllable aspect ratio. Chemical Communications, (7), pp.617–618. https://doi.org/10.1039/b100521i
42. Philip, D., 2010. Green synthesis of gold and silver nanoparticles using Hibiscus rosa sinensis. Physica E: Low-dimensional Systems and Nanostructures, 42(5), pp.1417–1424. https://doi.org/10.1016/j.physe.2009.11.014
43. Kalishwaralal, K., Deepak, V., Pandian, S.R.K., Kottaisamy, M., BarathManiKanth, S., Kartikeyan, B. and Gurunathan, S., 2010. Biosynthesis of silver and gold nanoparticles using Bacillus licheniformis. Colloids and Surfaces B: Biointerfaces, 77(2), pp.257–262. https://doi.org/10.1016/j.colsurfb.2010.01.014
44. Shukla, R., Bansal, V., Chaudhary, M., Basu, A., Bhonde, R.R. and Sastry, M., 2005. Biocompatibility of gold nanoparticles and their endocytotic fate inside the cellular compartment: a microscopic overview. Langmuir, 21(23), pp.10644–10654. https://doi.org/10.1021/la0513712
45. Natarajan, K., Selvaraj, S. and Ramachandran, S., 2010. Microbial synthesis of silver nanoparticles using Bacillus licheniformis and their antimicrobial activity. Colloids and Surfaces B: Biointerfaces, 79(2), pp.349–353. https://doi.org/10.1016/j.colsurfb.2010.04.030
46. Jain, D., Kachhwaha, S., Jain, R. and Kothari, S.L., 2011. Green and rapid synthesis of antioxidant silver nanoparticles using Argemone mexicana leaf extract and their antimicrobial activity. Journal of Microscopy and Ultrastructure, 9(2), pp.49–57. https://doi.org/10.4103/jmu.jmu_55_20
47. Durán, N., Marcato, P.D., Alves, O.L., De Souza, G.I. and Esposito, E., 2005. Mechanistic aspects of biosynthesis of silver nanoparticles by several Fusarium oxysporum strains. Journal of Nanobiotechnology, 3(1), p.8. https://doi.org/10.1186/1477-3155-3-8
48. Bhainsa, K.C. and D’Souza, S.F., 2006. Extracellular biosynthesis of silver nanoparticles using the fungus Aspergillus fumigatus. Colloids and Surfaces B: Biointerfaces, 47(2), pp.160–164. https://doi.org/10.1016/j.colsurfb.2005.11.026
49. Dahoumane, S.A., Yéprémian, C., Djédiat, C., Coute, A. and Lacroix, G., 2016. Enhancement of gold nanoparticle biosynthesis by photo-activation of the microalga Chlorella vulgaris. Journal of Nanoparticle Research, 18(6), p.158. https://doi.org/10.1007/s11051-016-3458-0
50. Lee, S.W., Mao, C., Flynn, C.E. and Belcher, A.M., 2002. Ordering of quantum dots using genetically engineered viruses. Science, 296(5569), pp.892–895. https://doi.org/10.1126/science.1068054
51. Amendola, V. and Meneghetti, M., 2009. Laser ablation synthesis in solution and size manipulation of noble metal nanoparticles. Physical Chemistry Chemical Physics, 11(20), pp.3805–3821. https://doi.org/10.1039/B900654N
52. Kim, D., Jeong, S. and Moon, J., 2006. Synthesis of silver nanoparticles using hydroxyl functionalized ionic liquids and their application to inkjet printing. Nanotechnology, 17(16), pp.4019–4024. https://doi.org/10.1088/0957-4484/17/16/034
53. Mallick, K., Witcomb, M.J. and Scurrell, M.S., 2001. Silver nanoparticle embedded antimicrobial paints based on vegetable oil. Materials Chemistry and Physics, 70(1), pp.21–25. https://doi.org/10.1016/S0254-0584(00)00450-8
54. Tsuji, T., Hashimoto, M., Nishikawa, D. and Tsuji, M., 2005. Preparation of silver nanoparticles by laser ablation in solution: influence of laser wavelength on particle size. Applied Surface Science, 252(5), pp.5222–5226. https://doi.org/10.1016/j.apsusc.2005.08.144
55. Song, J.Y. and Kim, B.S., 2009. Rapid biological synthesis of silver nanoparticles using plant leaf extracts. Bioprocess and Biosystems Engineering, 32(1), pp.79–84. https://doi.org/10.1007/s00449-008-0224-6
56. Huang, X., Jain, P.K., El-Sayed, I.H. and El-Sayed, M.A., 2007. Gold nanoparticles: interesting optical properties and recent applications in cancer diagnostics and therapy. Nanomedicine, 2(5), pp.681–693. https://doi.org/10.2217/17435889.2.5.681
57. Grabar, K.C., Freeman, R.G., Hommer, M.B. and Natan, M.J., 1995. Preparation and characterization of Au colloid monolayers. Analytical Chemistry, 67(4), pp.735–743. https://doi.org/10.1021/ac00101a026
58. Jana, N.R., Gearheart, L. and Murphy, C.J., 2001. Wet chemical synthesis of high aspect ratio cylindrical gold nanorods. The Journal of Physical Chemistry B, 105(19), pp.4065–4067. https://doi.org/10.1021/jp0107964
59. Cheng, Y., Samia, A.C.S., Meyers, J.D., Panagopoulos, I., Fei, B. and Burda, C., 2008. Highly efficient drug delivery with gold nanoparticle vectors for in vivo photodynamic therapy of cancer. Journal of the American Chemical Society, 130(32), pp.10643–10647. https://doi.org/10.1021/ja802786s
60. Xie, J., Lee, S. and Chen, X., 2010. Nanoparticle-based theranostic agents. Advanced Drug Delivery Reviews, 62(11), pp.1064–1079. https://doi.org/10.1016/j.addr.2010.07.009
61. Prakash, A. and Sharma, A., 2020. Evaluation of drug loading and release behavior of silver nanoparticles functionalized with polymers. Journal of Applied Polymer Science, 137(47), p.49389. https://doi.org/10.1002/app.49389
62. Singh, R. and Lillard, J.W., 2009. Nanoparticle-based targeted drug delivery. Experimental and Molecular Pathology, 86(3), pp.215–223. https://doi.org/10.1016/j.yexmp.2008.12.004
63. Rengan, A.K., Jagtap, M., De, A. and Banerjee, R., 2014. Multifunctional gold coated thermoresponsive liposomes for multimodal imaging and photothermal therapy. Biomaterials, 35(26), pp.7488–7496. https://doi.org/10.1016/j.biomaterials.2014.05.049
64. Torchilin, V.P., 2007. Micellar nanocarriers: pharmaceutical perspectives. Pharmaceutical Research, 24(1), pp.1–16. https://doi.org/10.1007/s11095-006-9132-0
65. Zeng, F. and Allen, C., 2004. Functionalized micellar nanoparticles for drug delivery. Journal of Controlled Release, 98(2), pp.237–247. https://doi.org/10.1016/j.jconrel.2004.05.004
66. Lee, S.H., Jun, B.H., Lee, Y.W. and Jeong, D.H., 2019. Gold and silver nanoparticles: synthesis and functionalization for biomedical applications. Nanomaterials, 9(6), p.814. https://doi.org/10.3390/nano9060814
67. Lu, W., Wan, J., She, Z. and Jiang, X., 2012. Brain delivery property and accelerated blood clearance of cationic albumin conjugated pegylated nanoparticles. Journal of Controlled Release, 161(3), pp.937–944. https://doi.org/10.1016/j.jconrel.2012.05.024
68. Bhattacharya, R. and Mukherjee, P., 2008. Biological properties of "naked" metal nanoparticles. Advanced Drug Delivery Reviews, 60(11), pp.1289–1306. https://doi.org/10.1016/j.addr.2008.03.013
69. Jain, T.K., Reddy, M.K., Morales, M.A., Leslie-Pelecky, D.L. and Labhasetwar, V., 2008. Biodistribution, clearance, and biocompatibility of iron oxide magnetic nanoparticles in rats. Molecular Pharmaceutics, 5(2), pp.316–327. https://doi.org/10.1021/mp700128j
70. Gu, Y., Chen, L., Zhang, X., Wang, J. and Xu, B., 2021. Surface modification of silver nanoparticles to improve stability and reduce cytotoxicity. Materials Science and Engineering: C, 119, p.111551. https://doi.org/10.1016/j.msec.2020.111551
71. Dahoumane, S.A., Mechouet, M., Wijesekera, K., Filipe, C.D.M. and Sicard, C., 2016. Green synthesis of gold nanoparticles using algae: Mechanisms and applications. Nano Today, 11(5), pp.701–718. https://doi.org/10.1016/j.nantod.2016.09.002
72. Dreaden, E.C., Alkilany, A.M., Huang, X., Murphy, C.J. and El-Sayed, M.A., 2012. The golden age: gold nanoparticles for biomedicine. Chemical Society Reviews, 41(7), pp.2740–2779. https://doi.org/10.1039/C1CS15237H
73. Gupta, A. and Mumtaz, S., 2020. Controlled drug release through silver nanoparticles: Challenges and future perspectives. Materials Science and Engineering: C, 108, p.110411. https://doi.org/10.1016/j.msec.2019.110411
74. Higuchi, T., 1963. Mechanism of sustained-action medication. Theoretical analysis of rate of release of solid drugs dispersed in solid matrices. Journal of Pharmaceutical Sciences, 52(12), pp.1145–1149. https://doi.org/10.1002/jps.2600521210
75. Jain, S., Hirst, D.G. and O'Sullivan, J.M., 2012. Gold nanoparticles as novel agents for cancer therapy. The British Journal of Radiology, 85(1010), pp.101–113. https://doi.org/10.1259/bjr/59448833
76. Korsmeyer, R.W., Gurny, R., Doelker, E., Buri, P. and Peppas, N.A., 1983. Mechanisms of solute release from porous hydrophilic polymers. International Journal of Pharmaceutics, 15(1), pp.25–35. https://doi.org/10.1016/0378-5173(83)90064-9
77. Kumar, B., Smita, K., Cumbal, L. and Debut, A., 2016. Green synthesis of silver nanoparticles using Andean plant extracts for antimicrobial applications. Advanced Materials Letters, 7(6), pp.467–472. https://doi.org/10.5185/amlett.2016.6280
78. Mallick, K., Witcomb, M.J. and Scurrell, M.S., 2001. Self-assembly of silver nanoparticles in a polymer matrix. Journal of Materials Science, 36(3), pp.733–738. https://doi.org/10.1023/A:1004897803456
79. Natarajan, K., Selvaraj, S., Sivakumar, M., Kumar, M. and Anburaj, D., 2010. Biological synthesis of silver nanoparticles using Bacillus licheniformis and evaluation of their antibacterial activity. Colloids and Surfaces B: Biointerfaces, 79(2), pp.488–493. https://doi.org/10.1016/j.colsurfb.2010.04.022
80. Reddy, N.J., Vali, D.N., Rani, M. and Rani, S.S., 2014. Evaluation of antioxidant, antibacterial and cytotoxic effects of green synthesized silver nanoparticles by Piper longum fruit. Materials Science and Engineering: C, 34, pp.115–122. https://doi.org/10.1016/j.msec.2013.08.019
81. Sanvicens, N. and Marco, M.P., 2008. Multifunctional nanoparticles–properties and prospects for their use in human medicine. TrAC Trends in Analytical Chemistry, 27(6), pp. 532–540. https://doi.org/10.1016/j.trac.2008.03.007
82. Selvakannan, P.R., Mandal, S., Phadtare, S., Gole, A., Pasricha, R. and Sastry, M., 2004. Water-dispersible tryptophan-protected gold nanoparticles. Small, 1(5), pp. 506–510. https://doi.org/10.1002/smll.200400059
83. Tiwari, D.K., Behari, J. and Sen, P., 2008. Application of nanoparticles in waste water treatment. World Applied Sciences Journal, 3(3), pp.417–433.
84. Zhang, X.D., Wu, D., Shen, X., Liu, P.X., Yang, N., Zhao, B., Zhang, H., Sun, Y.M., Zhang, L.A. and Fan, F.Y., 2012. Size-dependent in vivo toxicity of PEG-coated gold nanoparticles. International Journal of Nanomedicine, 6, pp.2071–2081. https://doi.org/10.2147/IJN.S21657
85. Baram-Pinto, D., Shukla, S., Perkas, N., Gedanken, A. and Sarid, R., 2009. Inhibition of HSV-1 attachment, entry, and cell-to-cell spread by silver nanoparticles. Journal of Nanobiotechnology, 7(1), p.11. https://doi.org/10.1186/1477-3155-7-11
86. Boisselier, E. and Astruc, D., 2009. Gold nanoparticles in nanomedicine: preparations, imaging, diagnostics, therapies and toxicity. Chemical Society Reviews, 38(6), pp.1759–1782. https://doi.org/10.1039/b806051g
87. Dreaden, E.C., Mackey, M.A., Huang, X., Kang, B. and El-Sayed, M.A., 2011. Beating cancer in multiple ways using nanogold. Chemical Society Reviews, 40(7), pp.3391–3404. https://doi.org/10.1039/C0CS00231A
88. Ghosh, P., Han, G., De, M., Kim, C.K. and Rotello, V.M., 2008. Gold nanoparticles in delivery applications. Advanced Drug Delivery Reviews, 60(11), pp.1307–1315. https://doi.org/10.1016/j.addr.2008.03.016
89. Kim, J.S., Kuk, E., Yu, K.N., Kim, J.H., Park, S.J., Lee, H.J., Kim, S.H., Park, Y.K., Park, Y.H., Hwang, C.Y. and Kim, Y.K., 2007. Antimicrobial effects of silver nanoparticles. Nanomedicine: Nanotechnology, Biology and Medicine, 3(1), pp.95–101. https://doi.org/10.1016/j.nano.2006.12.001
90. Lee, Y.S., 2013. Silver nanoparticles: synthesis and application for nanomedicine. International Journal of Nanomedicine, 8(Suppl 1), pp.219–225. https://doi.org/10.2147/IJN.S40880
91. Li, X., Robinson, S.M., Gupta, A., Saha, K., Jiang, Z., Moyano, D.F., Sahar, A., Riley, M.A. and Rotello, V.M., 2014. Functional gold nanoparticles as potent antimicrobial agents against multi-drug-resistant bacteria. ACS Nano, 8(10), pp.10682–10686. https://doi.org/10.1021/nn5042625
92. Liu, Y., Li, D., Wang, H., Zou, Q., Li, Y. and Zhang, X., 2020. Gold nanoparticle-based platforms for cirRNA detection and therapy. Journal of Materials Chemistry B, 8(23), pp.5051–5063. https://doi.org/10.1039/D0TB00665A
93. Lu, W., Singh, A.K., Khan, S.A. and Senapati, D., 2010. Gold nanoparticle-based detection and photothermal therapy of cancer. Therapeutic Delivery, 1(5), pp.687–704. https://doi.org/10.4155/tde.10.55
94. Marambio-Jones, C. and Hoek, E.M.V., 2010. A review of the antibacterial effects of silver nanomaterials and potential implications for human health and the environment. Journal of Nanoparticle Research, 12(5), pp.1531–1551. https://doi.org/10.1007/s11051-010-9900-y
95. Nasrollahi, S.A., Dadashzadeh, S., Haeri, A., Khoshayand, M.R., Najafi, F. and Shahverdi, A.R., 2011. Antibacterial activity of silver nanoparticles on Staphylococcus aureus. Drug Research, 61(2), pp.62–65. https://doi.org/10.1055/s-0030-1269850
96. Patra, C.R., Bhattacharya, R., Wang, E., Katarya, A., Lau, J.S., Dutta, S. and Mukherjee, P., 2008. Targeted delivery of gemcitabine to pancreatic adenocarcinoma using cetuximab as a targeting agent. Cancer Research, 68(6), pp.1970–1978. https://doi.org/10.1158/0008-5472.CAN-07-2400
97. Rosi, N.L. and Mirkin, C.A., 2005. Nanostructures in biodiagnostics. Chemical Reviews, 105(4), pp.1547–1562. https://doi.org/10.1021/cr030067f
98. Shao, D., Li, J., Zheng, X., Pan, Y., Wang, Z., Zhang, M. and Chen, L., 2020. Bioinspired diselenide-bridged mesoporous silica nanoparticles for dual-responsive protein delivery. Advanced Materials, 32(2), p.1905445. https://doi.org/10.1002/adma.201905445
99. Singh, P., Kim, Y.J., Zhang, D. and Yang, D.C., 2016. Biological synthesis of nanoparticles from plants and microorganisms. Trends in Biotechnology, 34(7), pp.588–599. https://doi.org/10.1016/j.tibtech.2016.02.006
Author Information
Authors: Mahesh Kumar Posa¹ᵃ, Ravi Kumar Kota², V.T.Iswariya³, P.Moulika⁴, S.Gejalakshmi⁵, Jagdish Kumar Arun¹ᵇ, Seema Sharma⁶ , and Sampat Singh Tanwar⁶*, Ravi Kumar Kota², V.T.Iswariya³, P.Moulika⁴, S.Gejalakshmi⁵, Jagdish Kumar Arun¹ᵇ, Seema Sharma⁶ , and Sampat Singh Tanwar⁶*
1a. Department of Pharmacology, School of Pharmaceutical Sciences, Jaipur National University, Jagatpura, Jaipur, Rajasthan, India, posamaheshkumarbabu@gmail.com
2. Department of Pharmaceutics, Jawaharlal Nehru Technological University, Hyderabad, Telangana, India, Ravi445@gmail.com
3. Department of Pharmaceutics, CMR college of pharmacy, Kandlakoya, medchal, Hyderabad, India
4. Department of Pharmaceutics, Joginpally B R Pharmacy college (JNTU), pallemoulika@gmail.com
5. Faculty of Pharmacy Dr.M.G.R.Educational and Research Institute, gejampharm@gmail.com
1b. Faculty of Pharmaceutical Science and Nursing, Department of Pharmacy Vivekananda Global University Jagatpura, Jaipur, India, drjagdisharun@gmail.com
6. Department of Pharmacy, Shri Vaishnav Vidyapeeth Vishwavidyalaya, Indore, M.P, India, seemasharmapharm@gmail.com
Corresponding Author:
Sampat Singh Tanwar, Assistant Professor, Department of Pharmacy, Shri Vaishnav Vidyapeeth Vishwavidyalaya, Indore, M.P, India
Email: sampattanwar1999@gmail.com, Orcid id: 0009-0001-3368-8636







Comments